|
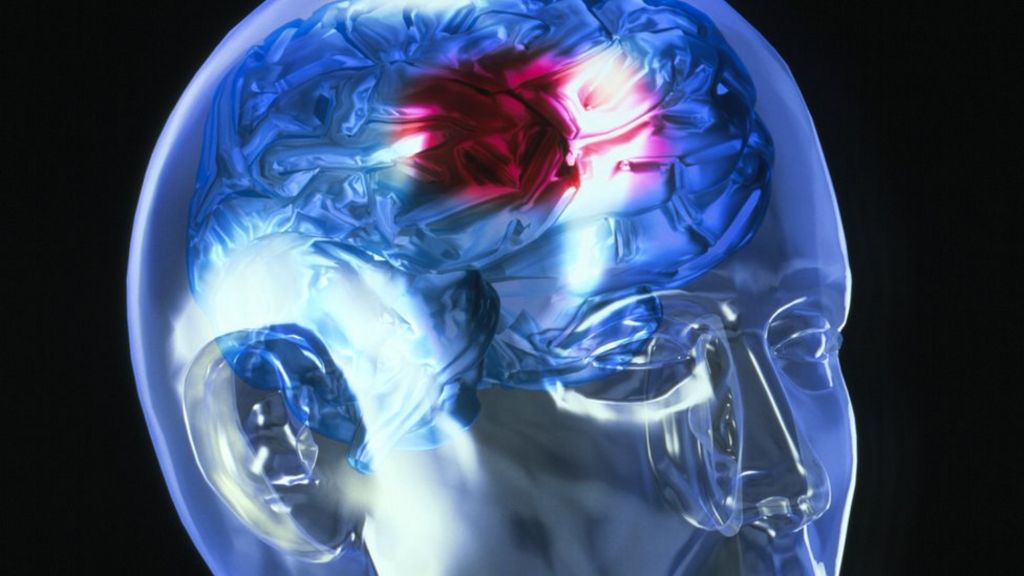
A meta-analysis of these trials concluded that rtPA given within 6 hours of a stroke significantly increased the odds of being alive and independent at final follow-up, particularly in patients treated within 3 hours. There have been 12 large scale, high-quality trials of rtPA in acute ischemic stroke. It can either be administered systemically, in the case of acute myocardial infarction, acute ischemic stroke, and most cases of acute massive pulmonary embolism, or administered through an arterial catheter directly to the site of occlusion in the case of peripheral arterial thrombi and thrombi in the proximal deep veins of the leg. The most common use is for ischemic stroke. TPA is used in some cases of diseases that feature blood clots, such as pulmonary embolism, myocardial infarction, and stroke, in a medical treatment called thrombolysis. The antidote for tPA in case of toxicity is aminocaproic acid. These drugs are used in clinical medicine to treat embolic or thrombotic stroke, but they are contraindicated and dangerous in cases of hemorrhagic stroke and head trauma. TPA can be manufactured using recombinant biotechnology techniques, producing types of recombinant tissue plasminogen activator ( rtPA) such as alteplase, reteplase, and tenecteplase. Human tPA is encoded by the PLAT gene, and has a molecular weight of ~70 kDa in the single-chain form. It is a serine protease ( EC 3.4.21.68) found on endothelial cells lining the blood vessels. 
It acts as an enzyme to convert plasminogen into its active form plasmin, the major enzyme responsible for clot breakdown. Tissue-type plasminogen activator, short name tPA, is a protein that facilitates the breakdown of blood clots.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
